Submitted and written by Dr. Bobby Nicholson, with peer review by Meyers and Bracey and major edits by Smith
84 yo F with hx of CAD presents to the ED after an out of hospital cardiac arrest. She arrives approximately 1 hr after arrest. She was out shopping with her daughter and began to feel unwell. She subsequently pressed her life alert and EMS was called to her location. On EMS arrival, she was pulseless. She received 2 rounds of CPR and 1 mg of epinephrine prior to obtaining ROSC. She was never defibrillated. Pre-hospital rhythm strips were not available; however, the reported arrest rhythm was PEA.
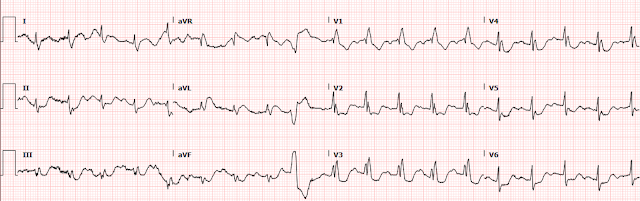
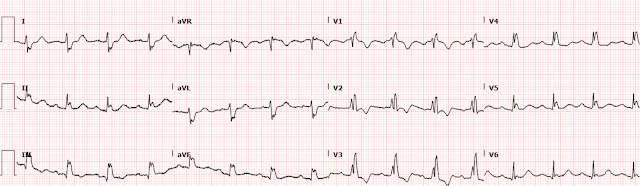
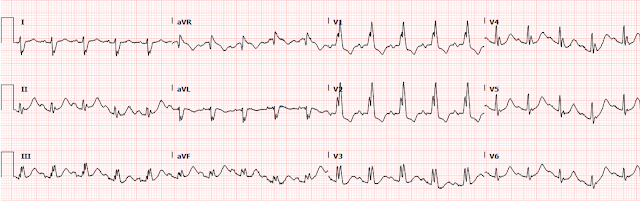
On arrival to the ED, she was Alert and Oriented x 4 and saturating 98% on 10L NRB. She was tachycardic to 110 bpm and her blood pressure was 110/74. EKG was immediately obtained:
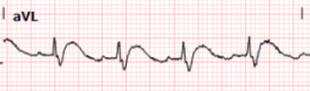
The rhythm appears to be sinus tachycardia with a RBBB and a single PVC noted. At the time of presentation, it was not known whether the patient had an existing RBBB. There also appears to be ST depression in most leads, which would not be surprising as one could expect a pattern of subendocardial ischemia after cardiac arrest.
However, if we look closer for OMI ECG findings:
1. there is also STD in the inferior leads with STE in lead aVL (maybe also lead I, but the baseline wander makes it hard to tell).
2. The T waves in aVL appear to be hyperacute.
3. There is STD in V1-V6, maximal in V3 and V4. There is still significant STD out to V6. aVR has a poor baseline and it is not totally clear how much STE is present there (the excessively discordant STE in the PVC in aVR suggest some STE).
Although there is tachycardia making the determination more difficult, the significant STD in V3-4 combined with the hyperacute T waves in aVL with reciprocal depression in III raised concerns for a posterolateral MI. We are publishing a study showing that STD maximal in V1-V4 is quite specific for posterior OMI, however we believe that the specificity decreases when there is tachycardia.
______
Smith additional comment: The fact of a PEA arrest makes ACS less likely than if it were a shockable arrest. When the first monitored rhythm in ACS arrest is not shockable, it is usually because the patient has been in VF so long that it has degenerated. However, when that is the case, the prolonged down time makes it much less likely that the patient will be awake at presentation. Pulmonary embolism is a quite common cause of PEA arrest, so one should always look closely at the right ventricle, even if the ECG looks like an OMI. I am uncertain if bedside cardiac echo was done here.
Why?
Cardiac arrest can lead to a severe type 2 OMI from supply demand mismatch (the low flow state causes such poor flow that there is subepicardial ischemia)
So it is possible that the ischemic ECG is due to cardiac arrest and tachycardia, and not due to ACS, but it must be assumed to be ACS (OMI) until proven otherwise by angiography.
According to these 2 randomized trials (TOMAHAWK and COACT, references at bottom), there is no utility of emergent angiography for cardiac arrest "without ST Segment elevation."
These studies did not address OMI ECG findings!!!
--There were many problems with the COACT trial. See my discussion at the bottom.
--And in the more recent TOMAHAWK, most patients were comatose and died of cerebral anoxia. So if the patient is awake and has ECG OMI findings, there is no reason to believe that angiography should be withheld.
____
The cath lab was activated, and the patient was treated
with aspirin and heparin. Cardiology evaluated the patient at the bedside. Patient was now
on NC and sitting upright in bed. Her only complaint is chest pain after CPR. The fact that she is alert and talking after supposed PEA arrest with multiple rounds of CPR likely indicates there may not have been full cardiac arrest but rather inability to feel pulses with very low but nonzero cardiac output. Ultrasound to detect cardiac activity and ultrasonographic pulses can be very helpful in these scenarios.
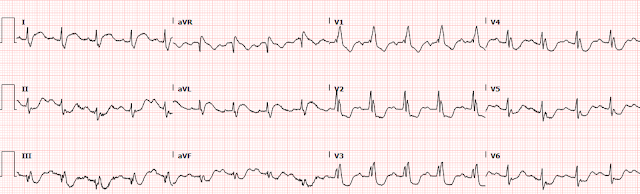
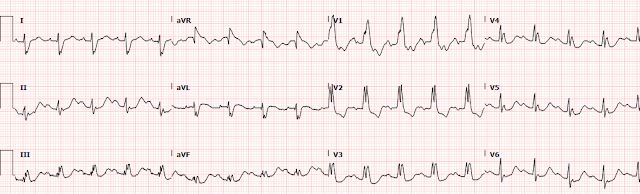
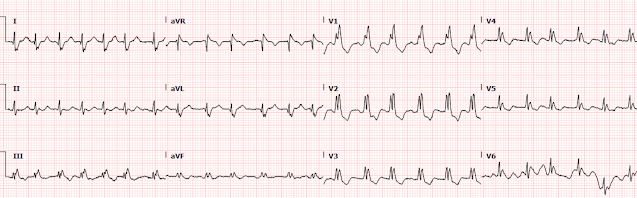
1254: repeat EKG with cardiology at bedside
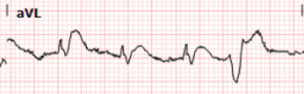
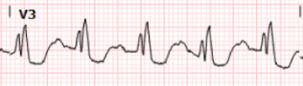
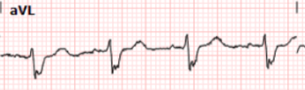
And here we have magnified leads aVL from both ECGs above:
Here we can clearly see that the T waves in aVL remain hyperacute appearing as they abut the end of the QRS complex with large area and volume, convex morphology, and have similar voltage as the R wave.
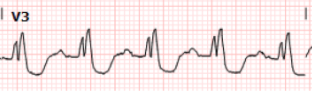
Next we have enlarged V3 for closer inspection:
When we compare V3, the ST depression is
slightly more pronounced, now approaching 3-4mm of depression. As previously
described, (http://hqmeded-ecg.blogspot.com/2018/02/right-bundle-branch-block-and-st.html)
V1-3 in RBBB can have some small, proportional STD at baseline, especially when there is an RSR’ pattern, however in this case the STD clearly exceeds normal proportions.
Thus, we have found excessively discordant STD maximal in V3-V4, and it must be considered diagnostic of posterior OMI until proven otherwise.
Patient was taken to the cath lab following cardiology evaluation. Review of her EMR revealed a prior history of CAD with remote history of CABG.
Her baseline EKG is below.
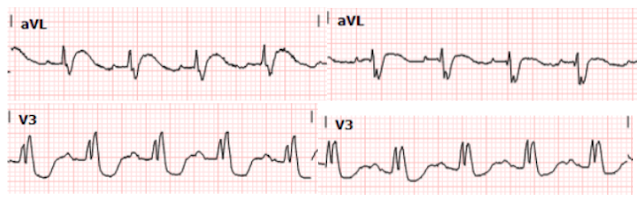
The change in morphology is quite staggering when placed side by side, as shown in close ups below:
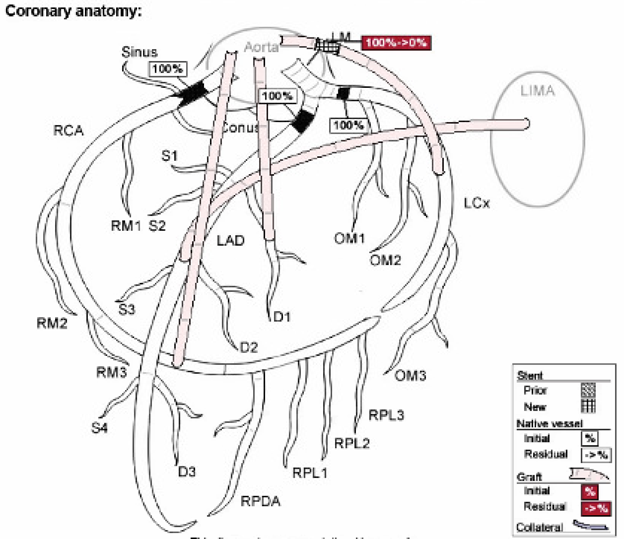
Cardiac catheterization revealed:
Complicated anatomy after prior CABG x 4 with
LIMA to LAD which was patent downstream. Her native RCA was occluded however
saphenous vein graft was patent to the distal right coronary circulation. The saphenous vein graft was also patent to the diagonal branch. Her native LAD and
circumflex were occluded, as was the saphenous
vein graft to the obtuse marginal which received balloon angioplasty
followed by aspiration thrombectomy without much improvement. A penumbra device
was then applied with injection downstream of nicardipine which restored flow
with an 80% lesion in the distal portion of the graft, to which a stent was
applied. This resulted in restoration of flow downstream of the OM circulation.
Thus, the culprit was total 100% (TIMI 0) acute thrombotic occlusion of the saphenous vein graft to the obtuse marginal (posterolateral territory). This fits the ECG findings perfectly.
Here is the first post-cath ECG at 1443:
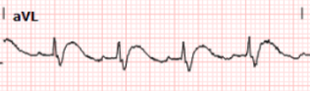
Post catheterization, we see normalization of
the patient’s T waves in I and aVL and deepening of the S wave. There is also
return towards her baseline ST depression in V2-3.
Below we show leads aVL and V3 before intervention (left images) and after (right images):
Here is the ECG later at 1922:
The latest ECG shows continued improvement.
Initial troponin (hs trop I) resulted after the patient was taken for catheterization, but was elevated at 2,598 ng/L. Repeat troponins during hospitalization were greater than 25,000 ng/L x 3. Her hospital course was complicated by cardiogenic shock requiring multiple pressors without improvement in her hemodynamics.
Echocardiography on hospital day 1 showed LVEF 55-60% with akinesis of the lateral wall segments and a small defect in the region between the normally contracting anterior wall and akinetic anterolateral segment. Additionally, there was echogenic material in the pericardial space concerning for organized heme. Despite being only day 1, this was interpreted as likely due to LV rupture with tamponade physiology.
Smith comment: there are often ECG findings of post-infarction regional pericarditis, which often precedes rupture. See this case: Cardiac Arrest, hypotension, tachycardia)
Cardiac MRI on hospital day 1 showed LVEF 45% with akinesis of the basal and mid anterolateral wall, basal inferior wall, and mid anterior wall. There was hyperkinesis of the septum, mid inferior wall, and apical LV regions. Again, the moderate sized heterogenous fluid collection in the lateral and inferior pericardial regions was noted. There was also a prominent collection immediately posterior to the basal anterolateral wall. Overall findings were most consistent with AMI related rupture of the basal anterolateral wall.
Ultimately, the patient’s family elected to
pursue palliation and conservative treatment. During hospitalization, she
developed pulmonary edema, encephalopathy, AKI, and paroxysmal atrial
fibrillation treated with IV amiodarone bolus and IV digoxin.
Hospital Day 2:
Despite conservative treatment, she progressed
rapidly during hospitalization and was weaned off all pressors with restoration
of her renal function and encephalopathy. Repeat echo showed LVEF 50 to 55%
with a questionable pericardial effusion. Patient was ultimately discharged to
acute rehab on hospital day 7.
Learning Points:
Like LBBB, paced rhythms, and all other abnormal and wide QRS complexes, RBBB follows the rules of appropriate discordance. This usually means there should be only a small, proportional amount of STD in the leads with the R', usually the right precordial leads.
In ACS, STD maximal in V1-V4 is posterior OMI until proven otherwise. In the setting of RBBB, this is STD that is proportionally excessive for the QRS complex.
Here is an example of an elderly person with complex known CAD who suffered cardiac arrest, did not have STEMI criteria on her post-arrest ECG, reported PEA arrest, who needed emergent reperfusion definitively based on her ECG findings. Many cardiologists in this situation would have denied her emergent reperfusion. I am honestly surprised that this patient received emergent catheterization. But I believe it was the right thing to do, and I believe her outcome would have been dramatically worse without reperfusion. We can do better than simply saying "no STEMI on post ROSC ECG? no cath."
Desch S, Freund A, Akin I, et al. TOMAHAWK. Angiography after Out-of-Hospital Cardiac Arrest without ST-Segment Elevation. N Engl J Med [Internet] 2021;Available from: http://dx.doi.org/10.1056/NEJMoa2101909
Lemkes JS, Janssens GN, van der Hoeven NW, et al. COACT. Coronary Angiography after Cardiac Arrest without ST-Segment Elevation. N Engl J Med [Internet] 2019;Available from: http://dx.doi.org/10.1056/NEJMoa1816897
I discuss COACT in this blog post, and paste that discussion here:
A woman in her 60s with VFib arrest and no STEMI on her post-ROSC ECG.
COACT Discussion, by Smith:
The COACT trial was fatally flawed, and now has many cardiologists convinced that if there are no STEMI criteria, the patient does not need to go to the cath lab.
Lemkes JS, Janssens GN, van der Hoeven NW, et al. Coronary Angiography after Cardiac Arrest without ST-Segment Elevation. N Engl J Med [Internet] 2019;Available from: http://dx.doi.org/10.1056/NEJMoa1816897
Should all patients with shockable arrest be taken to angiography regardless of STEMI or No STEMI?
There has long been controversy about whether to take patients with a shockable rhythm without ST Elevation to the cath lab, and a recent randomized trial showed no benefit: Coronary Angiography after Cardiac Arrest without ST-Segment Elevation (COACT). This study had a fatal flaw: they did not keep track of all the "Non-STEMI patients" who were NOT enrolled, but instead were sent for immediate angiogram. It was done in Europe, where the guidelines suggest taking all shockable arrests emergently to the cath lab. So it is highly likely that physicians were very reluctant to enroll patients whom they suspected had Occlusion MI (OMI), even if they did not have STEMI. These physicians did not want a patient with an OMI that was not a STEMI to be randomized to no angiogram. This strong suspicion is supported by their data: only 22 of 437 (5.0%) patients in this study had OMI.
What percent of shockable arrests without STE have an OMI?
This large registry in Circulation 2010 reported that at least 1 significant coronary artery lesion was found in 128 (96%) of 134 patients with ST-segment elevation on the ECG performed after the return of spontaneous circulation, and in 176 (58%) of 301 patients without ST-segment elevation.
5% vs. 58%!! It is clear that there was signficant enrollment bias in COACT.
We at Hennepin recently published this study
Sharma et al. (with Smith and others) found that among patients with shockable cardiac arrest who had OMI, the initial and subsequent pre-angiogram ECG were only 75% sensitive for OMI, with similar specificity.
Sharma A, Miranda DF, Rodin H, Bart BA, Smith SW, Shroff GR. Do not disregard the initial 12 lead ECG after out-of-hospital cardiac arrest: It predicts angiographic culprit despite metabolic abnormalities. Resuscitation Plus [Internet] 2020;4:100032. Available from: http://www.sciencedirect.com/science/article/pii/S2666520420300321

No comments:
Post a Comment
DEAR READER: I have loved receiving your comments, but I am no longer able to moderate them. Since the vast majority are SPAM, I need to moderate them all. Therefore, comments will rarely be published any more. So Sorry.