This case was provided by one of our fine 5th year EM/IM residents, Rachael Krob MD. The description of the method for inserting a pacing wire is by one of our fine 3rd year EM residents, Mark Sandefur MD.
Case
An elderly male was found on the floor with altered mental status. When EMS arrived, he was obtunded but occasionally able to answer yes/no questions. He was found to be bradycardic in the 20's-30’s. Glucose was normal.
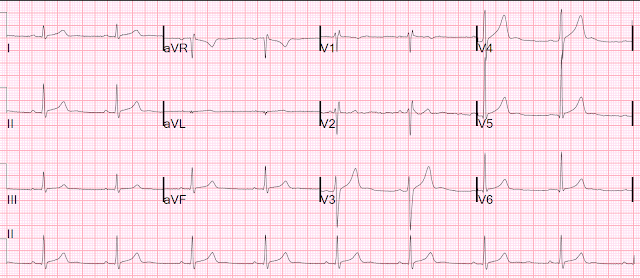
A prehospital 12-lead was recorded; unfortunately, only half of it made it into the chart:
The medics report that there was complete heart block. They gave atropine with no response, so they initiated transcutaneous pacing. They believed they had capture by palpating pulses and his mental status improved somewhat, so they had to give midazolam.
On arrival in the ED, he continued to have altered mental status and shock, and so was intubated using RSI.
A cardiac ultrasound was performed during transcutaneous pacing.:
A prehospital 12-lead was recorded; unfortunately, only half of it made it into the chart:
 |
| There is a slow indeterminate rhythm with a right bundle branch block morphology. |
The medics report that there was complete heart block. They gave atropine with no response, so they initiated transcutaneous pacing. They believed they had capture by palpating pulses and his mental status improved somewhat, so they had to give midazolam.
On arrival in the ED, he continued to have altered mental status and shock, and so was intubated using RSI.
A cardiac ultrasound was performed during transcutaneous pacing.:
You can see the atrium beating at a normal rate, but the ventricle is responding very slowly.
There is normal-appearing myocardial contractility, but that the transcutaneous pacing was not capturing.
Pacer pad placement was optimized with good anterior and posterior placement, and the amperage was turned all the way up, without capture. Because he was hypotensive and in shock, and he continued to have a heart rate in the 20-40’s, he was given 0.25 mg of push dose epinephrine, with improvement in blood pressure and heart rate.
Pacer pad placement was optimized with good anterior and posterior placement, and the amperage was turned all the way up, without capture. Because he was hypotensive and in shock, and he continued to have a heart rate in the 20-40’s, he was given 0.25 mg of push dose epinephrine, with improvement in blood pressure and heart rate.
The decision was made to place a transcutaneous pacing wire. While this was being placed, transcutaneous pacing was discontinued due to the failure of capture and quick response to low dose IV epinephrine. A 12-lead ECG was obtained:
This was interpreted as bradycardia with complete heart block. While the pacing wire was being inserted, the patient received push dose epinephrine every 3-5 minutes as his heart rate and blood pressure would drift down. During this period, continuous cardiac ultrasonography showed the pacing wire in the right atrium. There was some difficulty with advancing the wire through the tricuspid valve into the right ventricle, but eventually placement was successful and appropriate capture was achieved:
You can see the wire in the RV (see still picture with arrow in explanation below)
He was then admitted to the MICU with plans to go to the cath lab for a screw-in pacer. Outcome was good.
Placement of an Emergency Transvenous Pacer. Ultrasound is the preferred method.
Indications (1)
I. Used for
unstable bradycardia when other measures fail:
--Medical
treatment is inadequate (e.g.: atropine, treatment of hyperkalemia, reperfusion for ischemia, etc.)
--External (transcutaneous) pacing fails to achieve capture, even after optimal placement and high output, or when not tolerated by the patient
--Even if transcutaneous pacing succeeds, it is usually not a good solution for multiple hours and may not be persistently efficacious.
--Often utilized
in the setting of high grade AV node block, Sick Sinus Syndrome, etc.
II. Also can be
used for overdrive pacing for unstable tachydysrhythmias, especially Torsades de Pointes (polymorphic VT due to long QT).
Contraindications(1)
Prosthetic
tricuspid valve
Severe
hypothermia
--Predisposes
to VF
--Bradycardia is physiologic!
--Bradycardia is physiologic!
Studies
generally show approx 70-80% success rate.2,3
Average time to successful placement was 18 minutes.2
--30% less than 5
minutes
A 1981 study4
comparing flow directed balloon tipped catheters to standard semirigid
electrode catheters revealed several important differences:
--Improved
time: 6 min w/ balloon vs. 13 min w/ standard
--Fewer
complications with balloon tipped catheters
Most studies
assessing emergency transvenous cardiac pacing were preformed before the use of
ultrasound.
Techniques
There are a
couple well-described methods for this procedure:
--Sensing
method
--EKG
monitoring method
The main
benefit of these techniques was that a physician could determine when the
catheter reached the heart during the procedure without needing radiographs.
We will not discuss these techniques.
Ultrasound Technique: This is the preferred method
at Hennepin County Medical Center because it is technically simpler and there are fewer opportunities for
error. It utilizes another physician or sonographer for real time ultrasound
guidance.
Place a Sheath Introducer
Right internal jugular or left
subclavian veins preferred.
 |
| A Touhy-Borst Adaptor tightens around the wire, preventing leakage of blood or entry of air. |
Insert the Pacing Catheter:
Roughly measure distance that
will be needed.
Place a Tuohy-Borst adaptor with Cathgard
on the introducer.
Insert the temporary pacing catheter
into the Cathgard/sheath.
--Curvature must be directed to the
left so the catheter can enter the heart:
Insert with Ultrasound Guidance:
A second provider uses ultrasound to
identify the catheter entering the RV.
--Subcostal view preferred
The heart is bradycardic
Now the pacer wire is in place (see still image with arrow below) and the heart is being paced and is beating much faster.
The heart is bradycardic
Now the pacer wire is in place (see still image with arrow below) and the heart is being paced and is beating much faster.
Here is a still of the second ultrasound image:
 |
| The arrow shows the pacer wire. |
Pacing Module:
An assistant connects the pacing
catheter to the pacing module/box.
--Use the V ports
--Ensure correct placement of
positive and negative electrodes in the module
--When the catheter is seen in the
heart, turn on the pacing module.
Hit the “Emergency, Async”
button, which initiates
predetermined automatic settings (may vary depending on equipment):
--Asynchronized
--Rate 80
--A output 20 milliAmps (mA) (not applicable
for our purposes)
--V output 25 mA
Confirm capture: ultrasound,
pulse check, EKG
Adjust the rate if needed.
Tighten the Tuohy-Borst adaptor
to help prevent displacement and extend the sterile sleeve.
Final Details
Obtain a CXR
to confirm placement.
 |
| The pacer wire is in the RV. Difficult to see here. The image below shows its exact placement. |
 |
| Ideal placement is in the apex of the RV. This is not ideal, but is working perfectly. |
Further Management
Discuss need
for more definitive treatment with cardiology (placement of a screw-in
temporary catheter, permanent pacemaker, etc).
Temporary Pacers should be immediately replaced by more permanent ones. Without the fixation into the myocardium with a screw (as permanent ones have), the temporary pacers can come loose and this is very dangerous!
Admit the
patient to the ICU with pads in place.
References
1. Roberts JR. Roberts and Hedges’ clinical
procedures in emergency medicine, 6e. Philadelphis, PA: Saunders; 2014:278.
2. Rosenberg AS, Grossman JI, Escher DJW,
et al.: Bedside transvenous cardiac pacing. Am Heart J. 1969;77:697.
3. Kimball JT, Killip T: A simple bedside
method for transvenous intracardiac pacing. Am Heart J. 1965;70:35.
4. Lang R, David D, Herman HO, et al.: The
use of the balloon-tipped floating catheter in temporary transvenous cardiac
pacing. Pacing Clin Electrophysiol. 1981;4:491.