Written by Sean Trostel MD, with edits by Smith and Meyers
A man in his 80s with history of CAD, MI s/p
PCI, HTN, HLD, Afib, and HFrEF, presented with sudden onset, aching,
central chest pain radiating to the left arm and shortness of breath that began
in the morning, 1 hour prior to arrival to the ED.
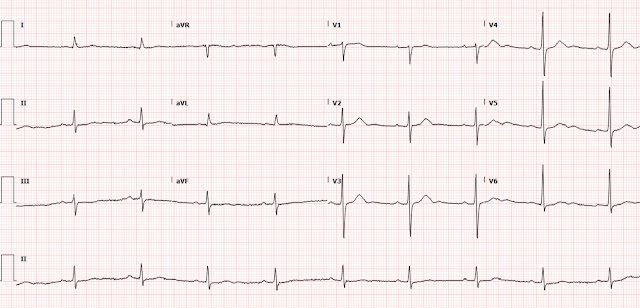
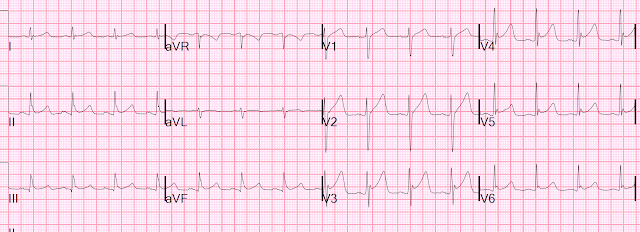
This was his initial triage ECG, recorded one hour after onset of symptoms, and a baseline ECG below for comparison:
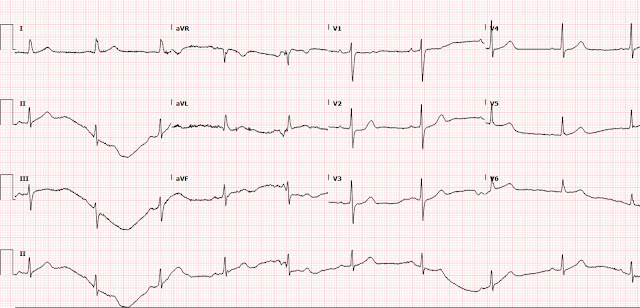 |
| What do you think? |
This ECG was interpreted as sinus bradycardia
with no acute ST or T wave changes compared to prior.
Despite the very poor quality and wandering baseline (and there was no evidence that it was repeated for better quality), the ECG is still diagnostic. There is subtle ST depression in the anterior
leads, most prominent in V2, which is specific for acute posterior OMI in the setting of ACS with a normal QRS complex (which should not have any STD). When compared to the prior ECG, the lateral T-waves have increased in volume
relative to the QRS, concerning for lateral involvement; this is partly because of a diminution of QRS amplitude. The ECG is diagnostic for posterolateral OMI until proven
otherwise.
The patient was observed in the ED, given
unspecified “pain medicine”, and labs were obtained. Initial high-sensitivity
troponin was elevated to 214 ng/L. Three hours later the patient continued to have chest
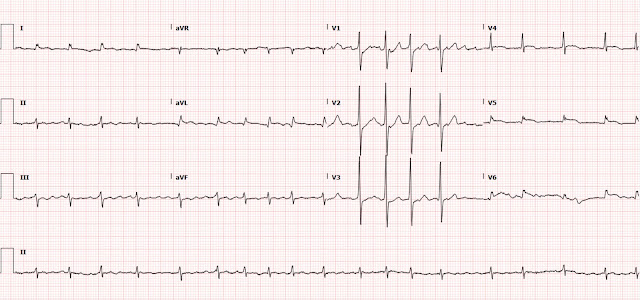
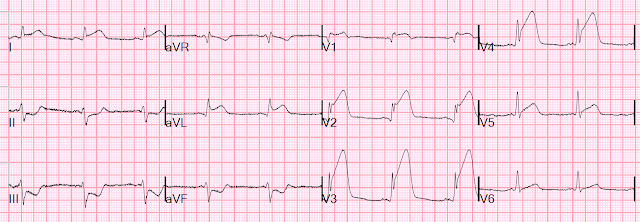
pain, and a repeat ECG was ordered:
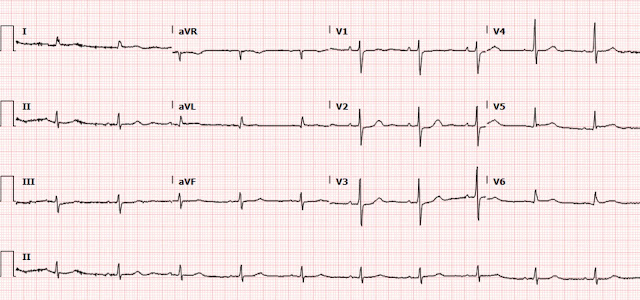 |
| Ongoing posterolateral OMI. |
At this time, the ED physician read the ECG to
have subtle lateral ST segment changes not appreciated on the prior ECG. But it seems that this was not acted upon.
A troponin was repeated and elevated to 1174 ng/L. The patient was transferred to a referral center, but the cath lab was not activated because his lateral ST elevation did not meet STEMI criteria. Neither the ED nor cardiology notes ever mention the ST depression seen in the anterior leads.
He was seen by cardiology on arrival to the referral
center, at which time he continued to have chest pain. He was diagnosed with
NSTEMI and was admitted for medical management and angiography the following
day.
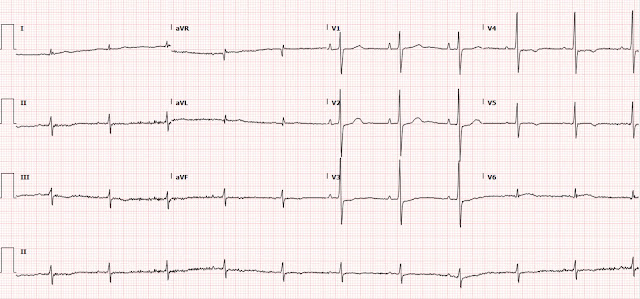
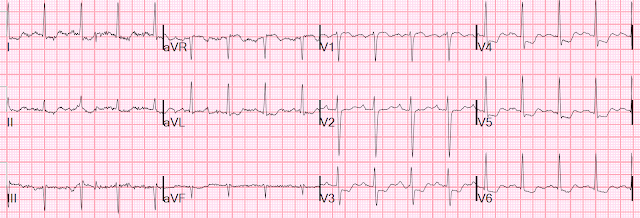
Overnight a serial ECG was obtained.
Did he develop Q waves and T-wave inversions
in the high lateral leads?
No, instead it is the classic limb lead reversal (LA/RA reversal). This causes inversion of lead I and switching of aVL for aVR and II for III. At the same time, the patient’s prior ischemic changes have improved. In the anterior leads you can see enlargement of the T-waves, likely reciprocal to T-wave inversions in the posterior leads, indicating some amount of reperfusion in the posterior myocardium. This is called "posterior reperfusion T waves."
Serial troponins continued to rise throughout
the night, 14,891 ng/L, then peaking at greater than 25,000 ng/L 12 hours after the onset of the patient’s pain.
The patient did undergo left heart cath approximately 24 hours after the onset of his pain, at which time an 80% mid RCA lesion and a 100% proximal first obtuse marginal (OM1) occlusion were found. The RCA lesion was called the culprit and stented, but they state there was no thrombus and give no detail about this lesion in the cath report. For the OM1 occlusion, there is literally no detail at all except "OM1: 100% stenosis." There is no detail of whether there is thrombus, no mention of it being a culprit lesion or chronic, no mention of any attempt to cross the lesion. There is no mention of collateral circulation to either territory.
Following PCI, an echo showed akinesis of the mid and apical anterior,
anterolateral, lateral, and inferior myocardium with LVEF 35-40% and no right
ventricular dysfunction. These changes were new compared to a prior echo. My
suspicion is that the OM1 occlusion could have been the true culprit lesion, given
posterolateral ischemia on ECG, akinesis in this region, and no RV involvement
on echo.
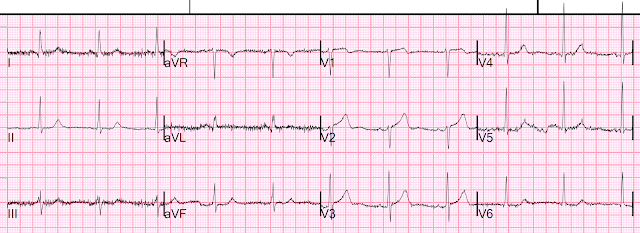
The following day, the patient developed Afib/flutter with RVR, and the following ECG was obtained.
In this ECG you can see recurrence and
worsening of the patient’s lateral ST elevation and anterior ST-depression.
This is not surprising if the OM1 was the culprit without revascularization, and now this territory has increased demand due to tachycardia.
Ultimately the patient was discharged after 3 days and followed up in clinic 1 week later, at which time the following ECG was obtained.
Compared to the patient’s initial ECG, his new baseline has developed new large R waves in the anterior leads, which is consistent with posterior infarction.
Sadly this patient spent the next several
months in and out of the hospital with heart failure exacerbations before dying
from the same.
Learning points:
Especially if you do not have expert ECG interpretation skills, do not settle for an ECG of such poor quality as the first ECG from this case! An interpreter with less experience could have recognized some abnormalities in that first ECG if it had better quality.
ST-depression of any amplitude maximal in V2-V4 in a patient with suspected ACS and without another QRS-based reason for STD should be considered posterior OMI until proven otherwise.(1) In this case, the ST depression was subtle, but ultimately proved diagnostic of a posterior OMI. This is yet another example of STEMI(-) OMI receiving delayed catheterization due to the current STEMI/NSTEMI paradigm.
It is often difficult to determine or prove the culprit artery even with the angiogram report. The report shows no evidence that the OM1 was even considered as a potential culprit lesion. Anecdotally, we find such problems very commonly with delayed NSTEMI catheterizations. What might have been an obvious acute culprit initially, is perhaps less obvious 24 hours later, and this mistake is facilitated by the cognitive bias that, "of course there's no total occlusion, that can't be the acute culprit, because the patient has only NSTEMI."
According to 2021 AHA/ACC guidelines, NSTEMI with refractory angina or hemodynamic or electrical instability is an indication for immediate cardiac catheterization and possible PCI.(2) This patient would have benefited from cath lab activation as soon as he arrived to the referral center with ongoing anginal chest pain and uptrending troponin, regardless of the presence or absence of ECG changes.
If an ECG shows an abrupt change in axis, inversion of lead I, and reversal of lead aVR with aVL and II with III, check for upper extremity limb lead reversal.
Prior posterior infarction can be recognized by prominent R waves in V1-V3. These R waves can be thought of as reciprocal to would-be Q waves in the posterior myocardium. (3,4)
Non-STEMI that is OMI has double the 1-year and 2-year mortality of Non-STEMI that is NOMI. (5)
References
1. Meyers HP, Bracey A, Lee D, Lichtenheld A, Li WJ, Singer DD, Rollins Z, Kane JA, Dodd KW, Meyers KE, Shroff GR, Singer AJ, Smith SW. Ischemic ST-Segment Depression Maximal in V1-V4 (Versus V5-V6) of Any Amplitude Is Specific for Occlusion Myocardial Infarction (Versus Nonocclusive Ischemia). J Am Heart Assoc. 2021 Dec 7;10(23):e022866. doi: 10.1161/JAHA.121.022866. Epub 2021 Nov 15. PMID: 34775811.
2. Lawton J, Tamis-Holland J, Bangalore S, Bates E, Beckie T, Bischoff J, Bittl J, Cohen M, DiMaio J, Don C, Fremes S, Gaudino M, Goldberger Z, Grant M, Jaswal J, Kurlansky P, Mehran R, Metkus T, Nnacheta L, Rao S, Sellke F, Sharma G, Yong C and Zwischenberger B (2021) 2021 ACC/AHA/SCAI Guideline for Coronary Artery Revascularization: Executive Summary: A Report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines, Circulation, 145:3, (e4-e17), Online publication date: 18-Jan-2022.
3. Perloff JK. The Recognition of Strictly Posterior Myocardial Infarction by Conventional Scalar Electrocardiography. Circulation. 1964 Nov;30:706-18. doi: 10.1161/01.cir.30.5.706. PMID: 14226169.
4. van Gorselen EO, Verheugt FW, Meursing BT, Oude Ophuis AJ. Posterior myocardial infarction: the dark side of the moon. Neth Heart J. 2007;15(1):16-21.
5. Khan et al. Impact of total occlusion of culprit artery in acute non-ST elevation myocardial infarction: a systematic review and meta-analysis. European Heart J, 2007. 38, 3082-3089.
===================================
MY Comment, by KEN GRAUER, MD (2/25/2022):
===================================
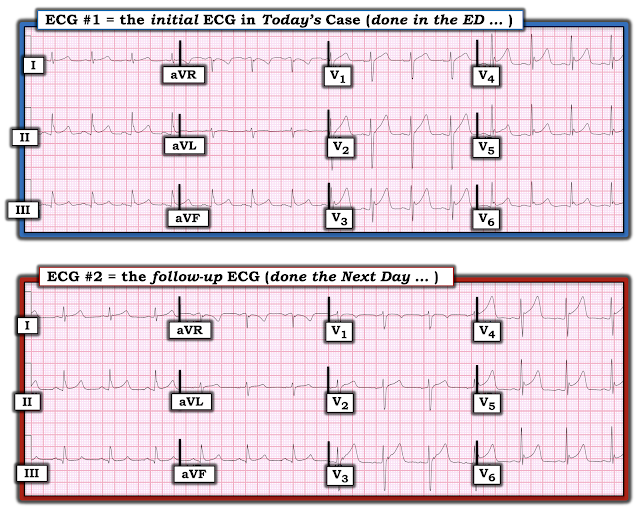
Excellent case by Drs. Trostel, Smith & Meyers — in which they present yet one more case of a posterior OMI that was overlooked by a number of providers despite the very typical history + progressively rising troponin levels. I limit my comments to the initial ECG — which I reproduce in Figure-1.
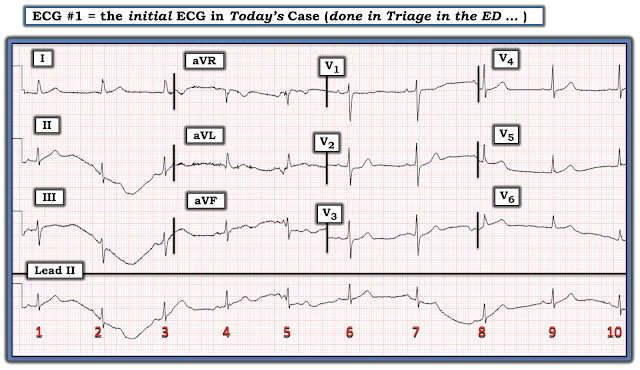 |
| Figure-1: The initial ECG in today's case, taken at Triage in the ED. |
My Thoughts on the Initial Tracing:
As per Dr. Trostel — there are subtle-but-real abnormalities on the initial tracing. These should have been noticed — and they should have triggered repeating the ECG long before the 2.5 hours it took.
- This patient is in his 80s — he has a history of known coronary disease — and he presents with a history completely typical for an acute event (in the form of new-onset, radiating chest pain that began just 1 hour before he got to the ED). With this history — Emergency providers need to start with an extremely high index of suspicion, to the point that they almost need to prove that such a patient is not having an OMI, rather than the other way around.
- Even before gaining access to the baseline (prior) ECG — the early transition in the chest leads (R = S, already by lead V2) with "shelf-like" ST depression in this lead V2 + the hint of ST elevation in lead V6 (the 1st complex in V6 looks to have a hyperacute T wave, in view of the very small amplitude of the R wave in this lead). Given the need to prove this highly typical acute presentation is not due to acute OMI — these changes (even before the elevated troponin levels returned) should not be ignored.
- Look at the baseline wander in the long lead II rhythm strip (Figure-2). The T waves in beats #1, 3 and 9 look hyperacute — where as it looks as if there is ST depression in beats #2, 5 and 7. In contrast — the ST-T wave of beat #6 looks totally normal.
- PEARL: The problem causing the marked baseline wander appears to be in the Left Leg — since everything looks normal in lead I ( = the 1 limb lead for which the electrical activity from the left leg electrode is not involved).
- In lead II — Is it the hyperacute-looking T wave (A) — or the marked ST depression in B?
- In lead aVL — Is it the ST elevation in B?
- In lead aVF — Is it the ST elevation in A — or the ST depression in B?
- in lead V2 — Is it complex A that is diagnostic of acute Posterior OMI (ie, with very positive "Mirror" Test) — or B, in which there is only subtle J-point depression? (NOTE: For more on the "Mirror" Test — See My Comment at the bottom of the page in the February 10, 2022 post in Dr. Smith's ECG Blog).
- In lead V3 — Is it the abnormal ST segment flattening in A (which in association with the "shelf-like" ST depression of A in lead V2 further supports the diagnosis of acute Posterior OMI) — or B in lead V3, which looks virtually normal?
- In lead V6 — Is it the hyperacute T wave with subtle-but-real ST elevation in A — or the unremarkable ST-T wave in B of lead V6?
- The technically inadequate initial ECG in Figure-1 should be immediately repeated in such a patient.
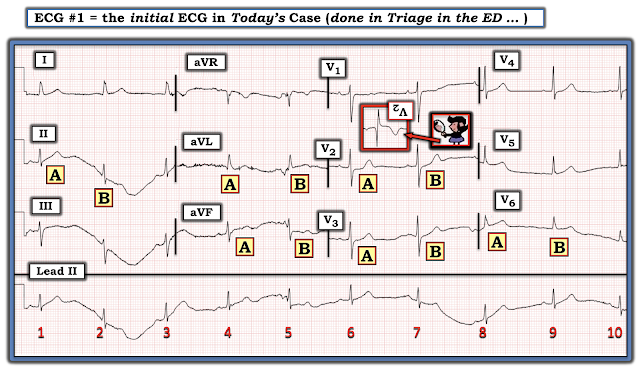 |
| Figure-2: I've labeled the initial ECG (See text). |